|
Other compounds are made from different combinations of atoms, like water… carbon dioxide… and table salt.Īnd that’s it! Nearly everything in this room, in your room, and in the entire universe, is made of: elements with one kind of atom, compounds containing different types of atoms chemically bonded together and mixtures of different elements and compounds together. Iron sulfide, a new substance, has been formed because the iron and sulfur atoms are now chemically bonded together. The iron can no longer be removed by a magnet because it’s bonded to the sulfur. atoms of the two elements are now joined together by chemical bonds. When the iron and sulfur are heated together… We can still separate the elements in the mixture. Protons, neutrons and electrons are the complete elements to form an atomic. The different elements are not joined together. Uses & Purposes of Atomic Structure Worksheet. This is now a mixture of the elements iron and sulfur. Homework Worksheet 3: Electron Configurations Write the full electron configuration for each of the following elements. If you know ONLY the following information can you ALWAYS determine what the element is (Yes/No) a. Atomic Structure Miss Adams Honors Chemistry 1 Name. 26 electrons, 29 neutrons, 26 protons 53 protons, 74 neutrons 2 electrons (neutral atoms) 20 protons 86 electrons, 125 neutrons, 82 protons O neutrons 11. When we mix two different pure substances together, like this, it’s a mixture. Name the element which has the following numbers of particles: a. It contains only sulfur atoms, and nothing else, so it is pure. 
This covers the structure of the atom, how to calculate protons, neutrons and electrons and why atoms have no charge. It is made of only one type of atom: iron atoms. Age range: 14-16 Resource type: Worksheet/Activity File previews docx, 40.07 KB docx, 15.5 KB docx, 20.68 KB pptx, 2.5 MB This is for low ability GCSE. Some substances, like particles of this iron, contain only one kind of atom. 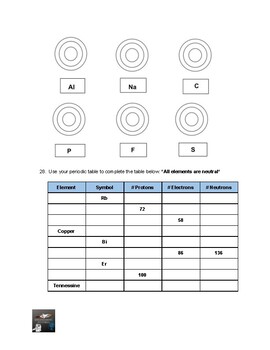
Describe the structure of a typical atom. Everything we can see and touch, and quite a lot that we can’t as well, is made of tiny particles called atoms. Describe how the modern atomic theory differs from these statements.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
